Isopropyl Alcohol vs. Hydrogen Peroxide: Benefits and Risks
Isopropyl Alcohol vs. Hydrogen Peroxide: Benefits and Risks
Isopropyl Alcohol vs. Hydrogen Peroxide: Benefits and Risks; did you know that people have used antiseptics since the 1800s? Yes, you read that right. Isopropyl alcohol and hydrogen peroxide were popular choices in cleaning contaminated surfaces and preventing the spread of germs.
Below, you’ll find out about the two solvents, how you can use them, and which is better than the other. So let’s dive right into the difference between hydrogen peroxide vs isopropyl alcohol.

What are Isopropyl Alcohol and Hydrogen Peroxide?
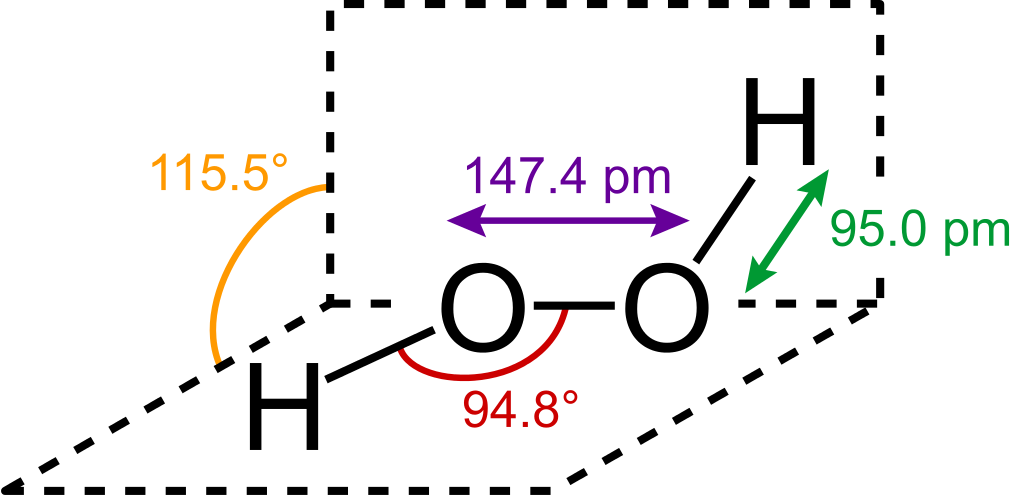
Isopropyl alcohol is a solvent with a molecular structure of three carbon atoms. Therefore, the disinfectant is also clear, colorless, and with an irritating smell. On the other hand, hydrogen peroxide has the molecular structure of H2O2, making it a highly potent oxidizer because of the two Oxygen atoms on its structure.
You can use both IPA and hydrogen peroxide for antiseptic purposes. However, there are differences in the speed with which each solvent is effective against microorganisms.
For example, isopropyl alcohol kills bacteria such as E.Coli and Staphylococcus in as little as ten seconds. Conversely, you’ll find that hydrogen peroxide is effective against certain viruses and bacteria, but the disinfectant requires five minutes or more to produce a favorable effect.
What are the Benefits of Using Isopropyl Alcohol?
Benefits of Using IPA
You can use rubbing alcohol in a hospital setting to clean the skin in surgical procedures or disinfect equipment such as thermometers. Isopropyl alcohol is an excellent cleaning and disinfecting agent, which can be used to cost-effectively clean environments like a laboratory. You can also use IPA to clean surfaces prone to contamination with bacteria and viruses.
Research shows that the solvent is effective against the COVID-19 virus. Therefore, use 70-95% Isopropyl alcohol for the best effect.
But you might ask, why not 100% alcohol?
That’s because 100% alcohol would coagulate the cell membrane proteins, thus forming a protective layer against which germs safeguard themselves, which you don’t want to happen.
Risks of using IPA; damage shellac, rubber, and plastic items, so be careful when cleaning objects containing those surfaces. Also, applying rubbing alcohol all over your skin is harmful since the solvent leads to itching, irritation, and rashes.
Your skin also absorbs IPA readily, leading to alcohol poisoning; therefore, use the solvent under an expert’s guidelines and keep it away from the reach of children.
What are the Benefits and Risks of Using Hydrogen Peroxide?
Benefits of Using Hydrogen Peroxide
Some people use hydrogen peroxide to clean minor cuts, wounds, and scrapes if they lack soap and water. And when grime contaminates your injury, bubbles from the solvent help remove the dirt, thus preventing an infection.
You can clean surfaces using 3% hydrogen peroxide but remember to rinse off the surface with water after application. Therefore, H2O2’s household utility includes cleaning your dishwasher, sink, and toilet. You can also treat plants with fungal infections. Or you can use the solvent to induce vomiting on a poisoned dog.
Risks of Using Hydrogen Peroxide
Avoid using hydrogen peroxide on large gaping wounds. That’s because the solvent irritates the skin and diminishes the body’s healing rate.
What Solvent is Better at Killing Germs?
Your preference for either isopropyl alcohol or hydrogen peroxide depends on factors such as; the surface you need to clean, the microbes you want to destroy, and how fast you need the effects.
Using a moderate amount of rubbing alcohol on the skin is safe, and that’s why surgeons prefer it. In contrast, hydrogen peroxide is irritating, and you shouldn’t use it on the skin.
The solvents are also effective on surfaces such as doorknobs, counters, and glass items. However, both IPA and H2O2 can destroy the finish on some objects, and it’s crucial to check product labeling before using any of them for cleaning purposes.
Experts recommend letting hydrogen peroxide sit for about 10 minutes on the surfaces you intend to clean for the solvent to provide the best results. So if you need a faster turnaround time, use rubbing alcohol instead.
How to Use Isopropyl Alcohol
- Purchase 70% IPA and don’t mix it with water as it’s already diluted.
- Then clean the item with water and soap.
- Next, apply the solvent evenly on the surface you intend to clean.
- Leave the object for 30 seconds before rinsing.
How to Use Hydrogen Peroxide
- Purchase 3% H2O2 but don’t dilute the solvent.
- Clean the object you desire to disinfect with soap and water.
- Then, apply hydrogen peroxide evenly using a piece of cloth.
- Leave the process for about 10 minutes for the best results.
Conclusion
Both Isopropyl Alcohol and Hydrogen Peroxide are effective as disinfectants and cleaning agents. People have used those solvents for over two centuries with excellent results. However, note that antiseptics irritate the skin, and you should use them sparingly with the help of a professional.
Both solvents provide similar effects as antimicrobial agents. However, use isopropyl alcohol if you’re looking for faster results. Also, IPA and H2O2 might destroy surfaces, so mind the items you’re cleaning.
